Out of thousands crowding store shelves, only a handful of dietary supplements have repeatedly shown clear health benefits in proper, controlled clinical trials. (7) American consumers spent $64.4 billion on supplements in 2023, but randomized research backs daily use for fewer than ten ingredients in the general population. The rest? Most end up matching placebo, while unbalanced diets or too little sunlight remain the real reasons people sometimes need supplements.
Evidence-based supplements earn their status through reproducible results, plausible biological mechanisms, and precise dosing tested in peer-reviewed trials. Vitamin D, fish oil, and vitamin B12 are rare standouts.
Most bestsellers – antioxidants, multivitamins – fail when put to a rigorous test. No pill erases the effects of a sedentary lifestyle or consistently poor nutrition. Study after study confirms that targeted supplementation only helps when there’s a clear deficiency or a specific medical issue.
The science draws a hard line between hope and outcome: few supplements are genuinely worth taking.
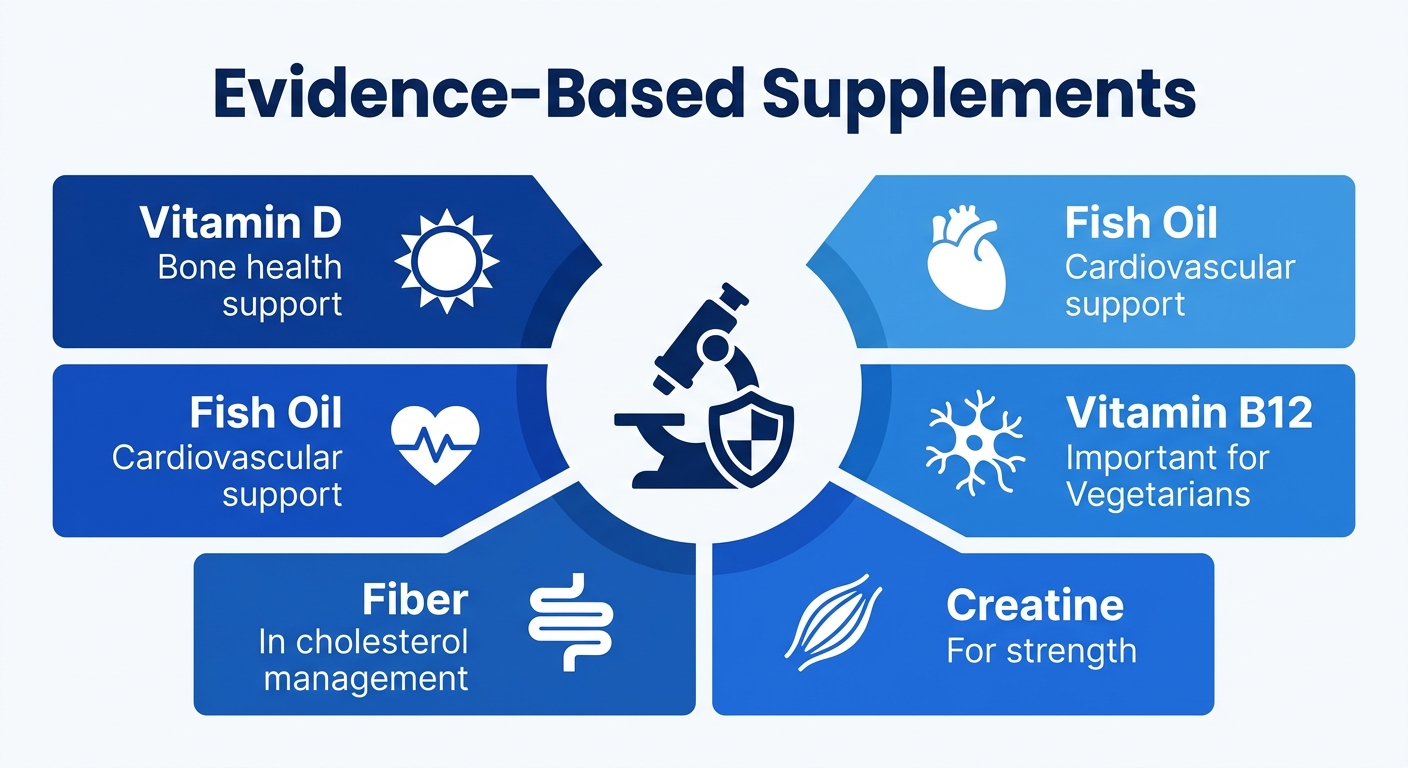
Top evidence-based supplements for general health
For older adults and strict vegetarians, Vitamin B12 supplementation prevents nerve damage and anemia – plant foods simply don’t supply bioavailable B12. (6)
Fish oil delivers omega-3s (EPA and DHA) that lower triglycerides and help prevent sudden cardiac death, especially for those who rarely eat fish.
Fiber supplements like psyllium lower LDL cholesterol and help control blood sugar, mainly in people who aren’t getting enough fiber from their diets.
Vitamin D – produced with sunlight – can prevent bone loss linked to deficiency in groups with little sun exposure or absorption issues.
Over 1 billion people globally have insufficient cholecalciferol levels.
Creatine monohydrate is one of the few ergogenic aids with strong support: the International Society of Sports Nutrition backs its use for better performance and lean mass gain in adults. No other supplement in its category comes close by evidence.
| Supplement | Recommended Population | Primary Benefit |
|---|---|---|
| Vitamin D | People with limited sun exposure, older adults | Prevents bone loss, supports immune function |
| Fish Oil (Omega-3) | Individuals not eating fish, cardiovascular risk patients | Reduces triglycerides, supports heart health |
| Vitamin B12 | Strict vegetarians, adults over 50 | Prevents anemia, supports nerve function |
| Fiber (e.g., Psyllium) | Low dietary fiber intake, metabolic syndrome | Lowers cholesterol, improves digestive health |
| Creatine Monohydrate | Active adults, older adults with muscle loss | Enhances muscle strength, improves exercise performance |
The evidence-based shortlist for general health in at-risk groups is just five: Vitamin D, fish oil, Vitamin B12, fiber, and creatine monohydrate.
Supplements with strong evidence vs overhyped supplements
By 2023, melatonin gummies had overtaken pharmacy aisles – but an NCCIH-cited review found 22 out of 25 brands misstated their ingredient content. (5)
Antioxidant supplements, once called disease shields, didn’t prevent cancer or heart attacks in major trials; in fact, beta-carotene raised lung cancer rates for smokers.
Multivitamins, taken daily by tens of millions, offered no protection against cardiovascular disease, cancer, memory loss, or death in meta-analyses with hundreds of thousands of participants.
Selenium was once pushed for cancer protection, but new studies tied it to higher diabetes risk – and no benefit for cancer prevention.
Beta-carotene supplements, once recommended for prevention, actually increased lung cancer rates in smokers.
Only one major supplement group – AREDS-formulated antioxidants – showed any real benefit: the NIH Age-Related Eye Disease Study found a 25% drop in risk of advanced macular degeneration with a specific vitamin-mineral blend.
Probiotics sometimes help with gut disorders, but there’s little reliable evidence they prevent problems in healthy adults.
“Of 25 melatonin gummy products tested in 2023, 88% failed label accuracy – one contained 347% of the stated dose.” – JAMA, NCCIH overview
Just one combination – the AREDS formula for macular degeneration – has strong evidence for disease prevention from a large randomized trial.
Cost-effectiveness of evidence-based supplements
In rural Bangladesh, pediatricians hand out zinc blister packs for children with acute diarrhoea. The World Health Organization supports this: a course of 20 mg zinc for 10 – 14 days shortens both illness time and severity, slashing hospital trips and deaths. Zinc runs less than three cents per day in bulk – far cheaper than IV fluids or long hospital stays.
This is one of the most cost-effective treatments for children under five.
In the United States, routine prenatal visits include folic acid supplements for anyone planning pregnancy. After reviewing rates of neural tube defects, the U.S. Preventive Services Task Force requires daily folic acid for all who could become pregnant. Neural tube defects, like spina bifida and anencephaly, mean lifelong care and surgery – sometimes costing six figures. A year’s supply of folic acid is less than a single day in neonatal intensive care.
Some supplements cost pennies per dose but avert medical bills running into the hundreds of thousands.
For adults with frequent migraine, doctors prescribe 400 – 500 mg magnesium oxide daily. The American Headache Society points to randomized trials showing fewer and less severe attacks. A year’s magnesium supply costs less than two triptan tablets and helps prevent dozens of migraine days.
Psyllium fiber supplements lower LDL cholesterol and help blood sugar for those getting too little dietary fiber. Psyllium is cheap and can slow or reduce the need for statins or diabetes drugs in some people.
Supplements like zinc, folic acid, magnesium, and fiber deliver some of the lowest cost-per-outcome ratios seen in preventive medicine.
Multivitamins vs individual targeted supplements
Pharmacies and corner stores both stock multivitamin bottles – each mixing vitamin D, B12, folate, and a handful of trace elements.
These blends promise broad coverage, but their one-size-fits-all approach rarely matches the precision of a single-ingredient solution.
Controlled trials show no reduction in heart disease, cancer, or general mortality among healthy adults taking multivitamins, even though use is widespread in the U.S. and Europe.
Despite decades of use, multivitamins show no mortality benefit in large randomized trials.
Meanwhile, targeted supplements – like folic acid before pregnancy or B12 for strict vegetarians – prevent specific deficiencies with direct, measurable outcomes.
The CDC still recommends pregnant and breastfeeding women use a multivitamin with iodine and folic acid, acknowledging broad gaps in these nutrients. On June 21, 2022, the U.S. Preventive Services Task Force ended support for beta-carotene or vitamin E for chronic disease prevention, citing repeated trial failures and occasional harm.
| Supplement | Source | Primary Function | Outcome Evidence |
|---|---|---|---|
| Multivitamin | Combined (D, B12, folate, minerals) | General nutritional insurance | No mortality or disease prevention in RCTs |
| Vitamin D | Individual, or as part of multivitamin | Bone health, deficiency correction | Reduces fracture risk in deficient adults |
| Folic acid | Individual (prenatal), or multivitamin | Neural tube development | Prevents neural tube defects in pregnancy |
| Vitamin B12 | Individual or multivitamin | Neurological function, anemia prevention | Prevents irreversible neuropathy in deficiency |
Multivitamins combine important nutrients, but can’t match the targeted impact of evidence-based, single-ingredient supplementation.
Synthetic vs natural supplement forms
Back in 2006, the National Eye Institute kicked off the AREDS2 trial to compare natural and synthetic antioxidants for slowing age-related macular degeneration. (4)
Lutein and zeaxanthin, two carotenoids from marigold flowers, took the place of synthetic beta-carotene in the revised formula after high lung cancer rates showed up in smokers using the synthetic version.
The NIH study followed over 4,200 participants for five years, tracking vision outcomes.
Synthetic beta-carotene increased lung cancer risk, while natural lutein and zeaxanthin improved eye health.
The analysis showed natural lutein plus zeaxanthin gave equal or better protection against advanced AMD than beta-carotene, which, ironically, raised lung cancer risk in high-risk groups. Beta-carotene – often made synthetically for supplements – underperformed not just because of its origin, but because isolated, high-dose synthetic forms can behave unpredictably in the body.
AREDS2 proved that natural-source carotenoids outclass synthetic ones in safety and impact when dosed precisely for a specific clinical goal.
Optimal dosing and timing of supplements
In 2024, new Endocrine Society guidelines made it clear: healthy adults under 75 usually don’t need vitamin D or calcium supplements. (3)
The U.S. Preventive Services Task Force reviewed results from 11 randomized trials with 51,419 adults over 50 and advised against both nutrients for routine fracture prevention.
JoAnn Manson, who led the VITAL trial at Harvard, confirmed that neither cholecalciferol nor calcium reduced major fracture risk in the general population.
Routine supplementation with cholecalciferol and calcium does not prevent fractures in healthy older adults.
Overdosing vitamin D – often more than 2,000 IU daily in over-the-counter products – brings risks: hypercalcemia, kidney stones, and vascular calcification have all been reported.
When to take supplements matters less than getting the dose right, and that remains the key factor for benefit and safety.
Leading medical guidelines now recommend supplements only in proven deficiency or for people at high risk – not as a blanket preventive for healthy adults.
Understanding what makes a supplement evidence-based
Surgeons at Edinburgh’s Royal Infirmary reversed scurvy with citrus juice in 1753-a milestone for supplement science.
Today, randomized clinical trials are the backbone of credible supplement evidence: only nutrients that consistently beat placebo in controlled settings get the “evidence-based” label.
Diseases like scurvy and rickets disappeared from Western hospitals after vitamin C and D were added to protocols, yet most modern supplement marketing can’t meet those clinical standards.
Efficacy is built on three things: benefit in randomized trials, a clear biological mechanism, and identical dosing across studies.
Randomized trial design – where participants are assigned by chance to supplement or placebo – controls for bias, natural recovery, and confounders, giving results that hold up across different populations.
By 1994, the U.S. supplement market had already outpaced regulatory science. Only ingredients with multiple, well-designed controlled trials-like cobalamin for anemia-meet the real evidence-based bar. Most supplements lack this: they’re sold on anecdote or assumption, not outcome data.
Randomized clinical trials remain the gold standard-they isolate cause and effect better than any other method, cutting through noise and wishful thinking.
Supplements with true evidence stand at a narrow crossroads: proven need, plausible mechanism, and repeated results – far fewer than most product labels dare suggest.
How does one determine the need for supplements and evaluate the supporting research?
In a Boston clinic, a patient after bariatric surgery brings her cyanocobalamin bottle – her surgeon ordered lifelong vitamin B12. Bariatric surgery alters the gut, blocking intrinsic factor and hindering absorption of B12, iron, and fat-soluble vitamins.
Without supplements, conditions like pernicious anemia or osteomalacia can appear within months.
Strict vegetarian diets, by cutting out animal foods, also eliminate bioavailable B12 – deficiency is guaranteed without fortified foods or supplements.
Pregnancy is a wildcard for metabolism: the American Thyroid Association and American Academy of Pediatrics both endorse daily iodine and folate to support fetal development and maternal thyroid health. A balanced diet covers most needs, but some life stages and medical interventions override those rules.
Deciding on supplements starts with diagnosis or risk assessment. Doctors check for deficiency with lab tests-serum cobalamin under 200 pg/mL, or 25-hydroxycholecalciferol below 20 ng/mL, for example-before recommending anything. Guessing based on symptoms or marketing leads to missed diagnoses, overuse, or dangerous combinations.
NIH pregnancy guidance points out that outside folate and iodine, major societies rarely recommend routine supplements for healthy adults. Population-wide vitamin regimens remain unsupported for most.
The quality of research sorts genuine evidence from hype. Trustworthy studies randomize people, use placebos, and track both good and bad outcomes. Anecdotes, animal data, or “surrogate” endpoints (like boosting blood levels with no clinical effect) don’t count. Peer-reviewed meta-analyses, not influencer stories, set the bar for real recommendations.
“Vegetarian, pregnant, or post-bariatric patients require supplements by necessity – not by trend or marketing.”
Medical history, lab tests, and real trials – not ad campaigns – decide who genuinely needs supplements.
How to research supplements using scientific sources?
In Baltimore, Larry Appel, M.D., scrolls through PubMed Clinical Queries – he types “magnesium supplement migraine,” restricting results to randomized trials. (2)
PubMed, from the National Library of Medicine, sorts through 36 million citations; Clinical Queries filters for robust trials, screening out anecdote and focusing on strong outcome data.
Nutrition teams at Johns Hopkins and Stanford also use this system to check for bias, sample size, and proper outcome measures before judging supplement risks or benefits.
USP and NSF International verify supplement identity, purity, and dosing – those logos on bottles mean independent lab checks. USP began in 1820 standardizing U.S. drug formulas; its protocols now support ingredient checks for thousands of supplements. NSF International, started in 1944, does blinded batch testing to catch adulteration or mislabeling before products hit shelves.
The NIH Office of Dietary Supplements creates fact sheets for both the public and clinicians, only listing supplements with real outcome data from controlled trials. Examine.com collects findings from randomized trials, cross-checking dose, population, and outcome for each supplement. These sources – unlike blogs or manufacturer sites – focus on peer-reviewed evidence and reveal funding or conflicts.
Systematic reviews and meta-analyses from Johns Hopkins or Stanford pool dozens of trials – filtering out weak endpoints and highlighting only reproducible results. Single-case stories or unsupported claims don’t survive their criteria. Researchers, not marketers, set the standard for “evidence-based.”
USP and NSF International, along with NIH and top university reviews, keep the supplement field tied to verifiable science.
Risks, misleading claims, and supplements to avoid
When Congress passed the Dietary Supplement Health and Education Act in 1994, manufacturers could suddenly launch new supplement ingredients without premarket safety testing or FDA approval. (1) Supplements got their own regulatory category, skipping the tough standards required for pharmaceuticals.
Pieter Cohen, M.D., an internist at Harvard, documented 23 cases of severe liver injury from “natural” herbal blends sold in U.S. stores – no FDA recall happened until hospitalizations rose. The FDA’s role is mostly reactive: warnings and recalls only come after reports of harm, not before.
Unlisted stimulants, synthetic analogues, and prescription drug contaminants have shown up in hundreds of supplements since 2007. NSF International and academic labs regularly spot these adulterants.
Dr. Fatima Stanford described a Massachusetts General patient hospitalized with tachycardia and high blood pressure – her “weight loss” supplement contained undeclared sibutramine, a banned drug.
John Cummings, M.D., notes that supplement-drug interactions – especially with anticoagulants, immunosuppressants, or chemotherapy – can cause life-threatening problems at seemingly low doses.
“Consumers trust that products on the shelf are safe, but the regulatory system assumes safety until proven otherwise. Only after adverse effects accumulate do authorities intervene.”
The FDA can’t require premarket proof of benefit or standardized dosing, so ingredient levels and purity often jump between batches. Dr. Cohen’s team found nearly one in five supplements tested included ingredients not listed on the label, and supplement-induced liver failure now makes up a growing share of acute hepatitis cases at U.S. poison control centers.
Risk is worse for people mixing several products, for children, and for those with chronic health issues.
Most supplements sold for “detox,” “rapid weight loss,” or “immune boosting” have no controlled trial evidence – these groups also account for most FDA warning letters and hospitalizations linked to supplement use.
Only products with verified ingredients, stable doses, and outcome data from randomized trials merit long-term use under a doctor’s supervision.
The FDA’s limited reach and the ease of launching new products keep untested, mislabelled, or even dangerous supplements on the U.S. market.
Sources
- FDA. NIH Office of Dietary Supplements text of DSHEA and FDA/USP legislative summary (1994).
ods.od.nih.gov - PubMed. National Library of Medicine PubMed Office Hours page.
nlm.nih.gov - Endocrine Society. 2024 vitamin D guideline and USPSTF draft recommendation on vitamin… (2024).
endocrine.org - NIH. National Eye Institute page on AREDS2 and related NIH reporting (2006).
nei.nih.gov - PubMed. JAMA research letter indexed in PubMed, ‘Quantity of Melatonin and CBD in… (2023).
pubmed.ncbi.nlm.nih.gov - NIH. Office of Dietary Supplements Vitamin B12 Fact Sheet for Consumers.
ods.od.nih.gov - Nutrition Business Journal. Market estimate cited in industry reporting (2023).
nai-online.com
F.A.Q
Which supplements have strong evidence for general health benefits?
Supplements with strong evidence include vitamin D, fish oil (omega-3s), vitamin B12, fiber such as psyllium, and creatine monohydrate. These are most beneficial for people with specific deficiencies or needs.
Who should consider taking vitamin B12 supplements?
Vitamin B12 supplements are important for strict vegetarians and adults over 50, since plant foods don't provide bioavailable B12. Deficiency can cause nerve damage or anemia.
Are multivitamins or antioxidant supplements worth taking?
Large studies show that daily multivitamins and most antioxidant supplements don't prevent disease or improve overall health for most people.
What are the risks of taking untested or mislabeled supplements?
Untested or mislabeled supplements may contain hidden ingredients, contaminants, or incorrect dosages. Such issues can lead to side effects, drug interactions, or serious health problems like liver injury.
How can I check if a supplement is evidence-based and safe?
Third-party testing logos such as USP or NSF International indicate quality. Reliable information on controlled clinical trials is available through the NIH Office of Dietary Supplements or PubMed.
Is fish oil supplementation beneficial for everyone?
For those who rarely eat fish or have cardiovascular risk, fish oil provides omega-3s that lower triglycerides and may help prevent sudden cardiac death.
Can supplements replace a healthy diet and lifestyle?
A poor diet or lack of exercise can't be corrected with supplements. Targeted supplements are effective only when there's a clear deficiency or a specific medical need.