Antibiotic resistance caused at least 1.27 million deaths in 2019 and played a role in nearly 5 million global fatalities (5).
Alexander Fleming discovered penicillin in 1928, launching an era where it looked like medicine had finally outsmarted bacteria. That didn’t last. Within twenty years, hospitals were filled with cases of penicillin-resistant Staphylococcus aureus.
Every new antibiotic class – sulfonamides in the 1930s, tetracyclines in the 1940s, fluoroquinolones in the 1960s – was followed by resistance, as microbes simply adapted faster than chemists could keep up.
Bacteria outpaced innovation. The arc of antibiotic resistance is full of irony: every new drug bought only a brief victory before resistance took over. By 2019, antimicrobial resistance killed more people than HIV/AIDS or malaria.
What is antibiotic resistance and how is it defined?
Back in 1940, Abraham and Chain found beta-lactamase – an enzyme that neutralizes penicillin – in a strain of Escherichia coli (4).
The Centers for Disease Control and Prevention (CDC) now say antibiotic resistance is when bacteria survive, or even grow, despite being hit with drugs meant to kill them.
The World Health Organization (WHO) uses “antibiotic resistance” only for bacteria – not viruses, not fungi – and calls antibiotics medicines for treating or preventing bacterial infections.
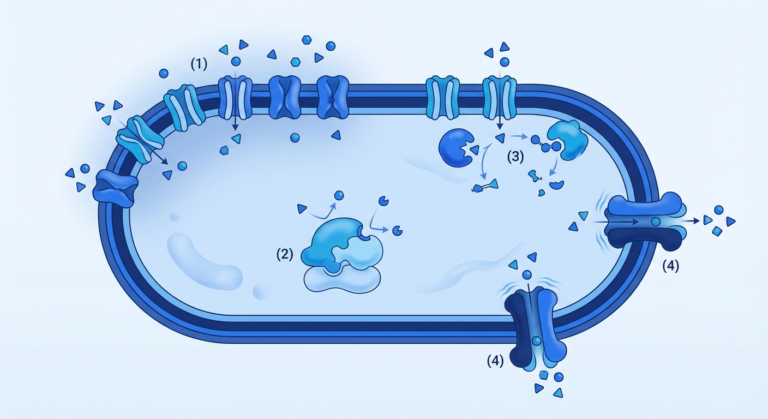
Bacteria don’t stick to one trick: they produce enzymes like beta-lactamase, pump drugs out using efflux pumps, or change the shape of their drug targets, making antibiotics useless.
Plasmids loaded with resistance genes travel between cells on mobile genetic elements, spreading resistance sideways – within and between species. A single resistant bacterium can multiply and create a population immune to an entire class of antibiotics.
One resistant bacterium can spawn a population immune to an entire antibiotic class.
Beta-lactamase, efflux pumps, and mobile genetic elements have become the main molecular weapons bacteria use to break new antibiotics.
When and how was antibiotic resistance first discovered?
Already in 1929, Alexander Fleming noted in the British Journal of Experimental Pathology that Bacillus coli could survive penicillin.
He watched colonies persist right at the drug’s edge, hinting that poor dosing let some bacteria escape.
Even decades earlier, Paul Ehrlich had seen trypanosomes become resistant to arsenicals, which led to his famous claim: “no chemotherapy without resistance.”
By 1940, Edward Abraham and Ernst Chain had pulled penicillinase out of Escherichia coli, proving bacteria could shut down penicillin with enzymes.
By 1942, hospitals were seeing penicillin-resistant Staphylococcus aureus infections – resistance had moved from the lab to the bedside.
Sulfa drugs, used before penicillin, had already run into resistance from Streptococcus and Neisseria in the 1930s. Still, enzyme-driven destruction of penicillin by E. coli marked a new chapter in resistance.
Penicillin resistance was documented in hospitals within three years of penicillin’s first clinical use.
In his 1945 Nobel lecture, Fleming warned that careless use of antibiotics would breed stronger bacteria.
The first person given penicillin, Albert Alexander, actually died because there wasn’t enough drug, not because of resistance – an odd prelude to the era when resistance would rule clinical outcomes.
Hospitals in the early 1940s quickly became the first places where evolution outpaced medical progress.
Bacteria with penicillinase showed that antibiotic resistance was just natural selection at work.
Key milestones in the history of antibiotic resistance from the 20th century to today
1961, London hospital lab: Patricia Jevons looked at Staphylococcus aureus that methicillin couldn’t kill – the antibiotic made for tackling penicillin resistance (3).
Methicillin-resistant Staphylococcus aureus (MRSA) appeared in less than two years after methicillin’s rollout, exposing a pattern: every new drug was met by a new adaptation.
By 2005, CDC counted 94,360 invasive MRSA cases and 18,650 hospital deaths in the U.S., turning MRSA from a lab surprise into an epidemic.
Vancomycin, once the last hope, didn’t last either: vancomycin-resistant enterococci were found in Europe in the late 1980s, with vancomycin-resistant Staphylococcus aureus following in the U.S. in 2002.
Each new “last-resort” antibiotic has faced resistance within just a few years of its introduction.
The 2008 discovery of New Delhi metallo-beta-lactamase 1 (NDM-1) in Klebsiella pneumoniae marked the global leap of carbapenem resistance, carried by plasmids, across countries and hospitals.
In 2013, the CDC officially classified threats as urgent, serious, or concerning. By 2015, the U.S. government produced the National Action Plan for Combating Antibiotic-Resistant Bacteria.
The WHO’s first expert meeting on resistance in 1959 is now just a faint echo in a crisis that’s fully global.
Each new antibiotic class – methicillin, vancomycin, carbapenems – triggered its own resistance story. The window of effective use shrank from decades to just a few years.
Key scientists and landmark studies that shaped our understanding of antibiotic resistance
Late 1940s, Hammersmith Hospital: Mary Barber isolated Staphylococcus strains defying penicillin, and resistance stopped being just a lab issue – it became an everyday clinical problem (2).
Barber’s records showed British hospital resistance rates jumping from almost nothing to over 50% by 1951, forcing hospitals to take infection control seriously.
In the U.S., Max Finland at Boston City Hospital tracked similar patterns, linking rising patient deaths to penicillin’s declining success. By 1956, resistance was a worldwide concern.
| Year | UK Hospitals: Penicillin Resistance Rate (%) | US Hospitals: Penicillin Resistance Rate (%) |
|---|---|---|
| 1946 | less than 5 | less than 5 |
| 1951 | 50 | 38 |
| 1956 | 59 | 56 |
Selman Waksman – who named “antibiotic” and found streptomycin – expanded the view past penicillin, showing resistance in soil microbes even before human use.
René Dubos traced resistance to ecological pressures, warning that it wasn’t only clinical misuse but environmental selection driving the trend.
Lawrence Garrod and Max Finland set up WHO’s first surveillance, standardizing lab methods so results were comparable worldwide.
In 1976, Stuart Levy ran a farm trial for the Alliance for the Prudent Use of Antibiotics: chickens fed tetracycline bred resistant E. coli, which passed to farm workers – proving agricultural use could spread resistance to people.
In a 2016 JAMA review, Thomas J. Walsh mapped resistance genomics, showing that mobile DNA elements allow resistance to jump between species and continents.
“We have propelled ourselves into a new era of infectious diseases – one where our own technology, deployed without foresight, now returns as a threat,” Stuart Levy warned in 1992.
Barber, Waksman, Dubos, and Levy each uncovered a different angle of resistance – hospital, lab, environment, agriculture – forcing medicine to face an adversary of its own making.
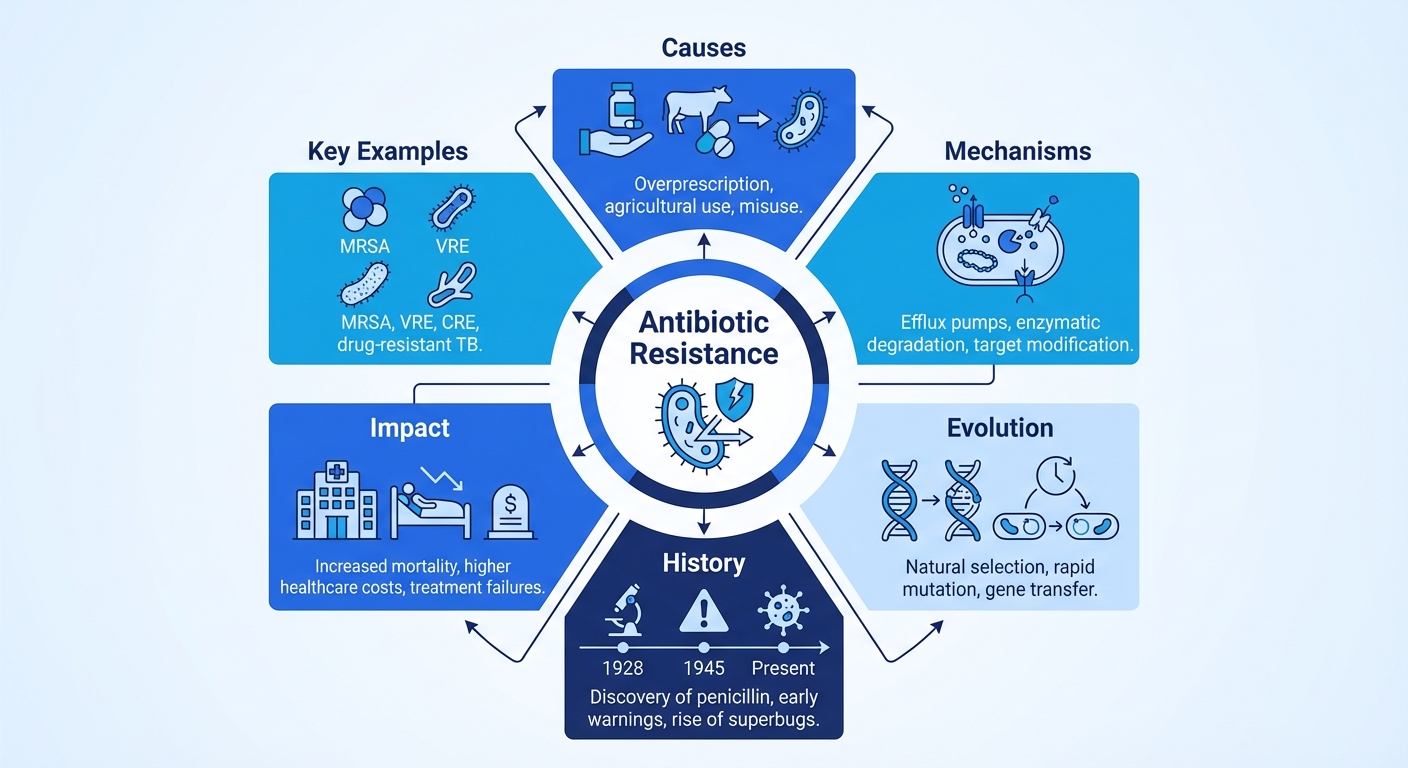
Causes of rising antibiotic resistance: overuse, misuse, and systemic failures
Indiscriminate antibiotic use – by doctors, farmers, and pharmaceutical companies – has changed the microbial world faster than any regulator could react.
In the early 1950s, agriculture started mixing tetracycline and penicillin into animal feed, making livestock grow faster but also creating reservoirs of resistant microbes in soil, water, and meat.
By 2016, a JAMA review had connected the rise in resistance to bad prescribing in both human and veterinary medicine, blaming family doctors and vets alongside big agriculture.
Drug companies pushed broad-spectrum drugs without precise dosing guides, saturating hospitals, farms, and homes with selection pressure.
Stewardship programs – audits, restrictions, education – came too late, letting multidrug-resistant microbes spread unchecked across regions.
Antibiotics meant to save livestock and patients ended up breeding resistance that spreads between farm, hospital, and home.
| Sector | Mechanism of Contribution | Impact of Resistance |
|---|---|---|
| Healthcare | Inappropriate prescribing, lack of stewardship | Therapeutic failures, increased mortality, prolonged hospital stays |
| Agriculture | Antibiotic growth promotion, environmental contamination | Resistant pathogens in food chain, ecosystem disruption |
| Veterinary | Routine prophylaxis, unregulated drug access | Zoonotic transfer, loss of effective animal treatments |
With no checks on overuse in medicine, agriculture, and veterinary practice, a global crisis took hold – bouncing between hospitals, farms, and kitchens.
Comparing eras and resistance trends: how the threat has evolved over time
By the late twentieth century, Gram-negative bacteria had replaced Gram-positives as the main drivers of drug resistance in U.S. hospitals (1).
Staphylococcus aureus sounded the first alarms, but soon Klebsiella pneumoniae, Pseudomonas aeruginosa, and Acinetobacter baumannii were dominating intensive care units packed with broad-spectrum drugs.
Plasmid-mediated resistance overtook point mutations as the go-to strategy: mobile genetic elements moved between Enterobacteriaceae, and carbapenem resistance spread in less than ten years.
In 2019, more than 2.8 million Americans got infections that standard antibiotics couldn’t treat, resulting in over 35,000 deaths per CDC data.
Gram-negative superbugs now spread resistance genes across species in years, not generations.
The “superbug” label moved from penicillin-insensitive Staphylococcus to Gram-negatives that shrug off multiple drugs, as resistance evolved from single-drug failures to pan-resistant strains.
Fungi, once rare in hospital charts, now cause outbreaks of pan-resistant Candida auris, echoing bacterial resistance but with their own twist.
Every era of antibiotics – sulfonamides, penicillins, cephalosporins, carbapenems – was outpaced by rapid evolution and gene swapping. Mobile genetic elements erased barriers between hospital, community, and farm.
Over the past seventy years, the signature of resistance has shifted from single-drug failures in isolated wards to global waves of multidrug-resistant “superbugs” fueled by lateral gene transfer.
| Pathogen Type | Primary Resistance Mechanisms | Therapeutic Impact |
|---|---|---|
| Gram-positive bacteria | Target modification, enzyme production (e.g., β-lactamase) | Loss of standard beta-lactam efficacy, MRSA epidemics |
| Gram-negative bacteria | Efflux pumps, porin mutations, plasmid-mediated enzymes (e.g., NDM-1) | Multidrug resistance, pan-resistance, ICU outbreaks |
| Fungi | Altered membrane sterols, efflux pumps, target modification | Azole and echinocandin failure, emergence of C. auris |
Now, Gram-negative bacteria set the pace for global resistance, their gene-swapping powers fueling the age of superbugs.
Sources
- CDC. ‘s Emerging Infectious Diseases review states that in the 1970s….
wwwnc.cdc.gov - mbevidencias.apm.org.br. Bibliographic record for Mary Barber’s article ‘Staphylococcal Infection due….
mbevidencias.apm.org.br - Patricia Jevons. Published ‘Celbenin-resistant Staphylococci’ in the British Medical Journal… (1961).
pmc.ncbi.nlm.nih.gov - CDC. Nature article on penicillinase history states that the presence of the… (1940).
nature.com - WHO. The Lancet systematic analysis of global bacterial antimicrobial resistance… (2019).
pubmed.ncbi.nlm.nih.gov
F.A.Q
What is antibiotic resistance and how does it happen?
Antibiotic resistance occurs when bacteria continue to grow or survive after treatment with antibiotics meant to kill them. They achieve this by producing enzymes, pumping drugs out, or altering the drug’s target.
When was antibiotic resistance first discovered?
The first observation of antibiotic resistance came from Alexander Fleming in 1929, when he noticed bacteria surviving penicillin. By 1940, scientists had identified an enzyme in E. coli that could destroy penicillin, proving resistance existed.
What are the main causes of rising antibiotic resistance?
The main drivers of resistance are overuse and misuse of antibiotics in medicine and agriculture. Inappropriate prescriptions and weak stewardship programs further increase the chances for resistant bacteria to thrive.
How do bacteria become resistant to antibiotics?
To counter antibiotics, bacteria produce enzymes like beta-lactamase that break down drugs, use efflux pumps to expel them, or modify the drug’s target. They also share resistance genes through mobile genetic elements such as plasmids.
Which antibiotics have faced resistance soon after their introduction?
Resistance has emerged for nearly every major antibiotic class soon after introduction. Penicillin resistance appeared within a few years, followed by resistance to sulfonamides, tetracyclines, methicillin, vancomycin, and carbapenems.
Why is antibiotic resistance considered a global health threat?
Infections become much more difficult or even impossible to treat due to antibiotic resistance. In recent years, it has caused more deaths than HIV/AIDS or malaria. It spreads rapidly in hospitals, farms, and communities, rendering many standard treatments ineffective.
Can antibiotic resistance spread between animals and humans?
Antibiotic resistance can transfer from animals to humans, particularly when antibiotics are used in agriculture. Resistant bacteria from livestock may spread to farm workers and enter the food supply.