In 1900, infectious diseases were the top killers, dragging life expectancy in industrialized countries down to 47 years. That changed in the 1940s, once penicillin made it possible to cure bacterial infections that were once a death sentence.
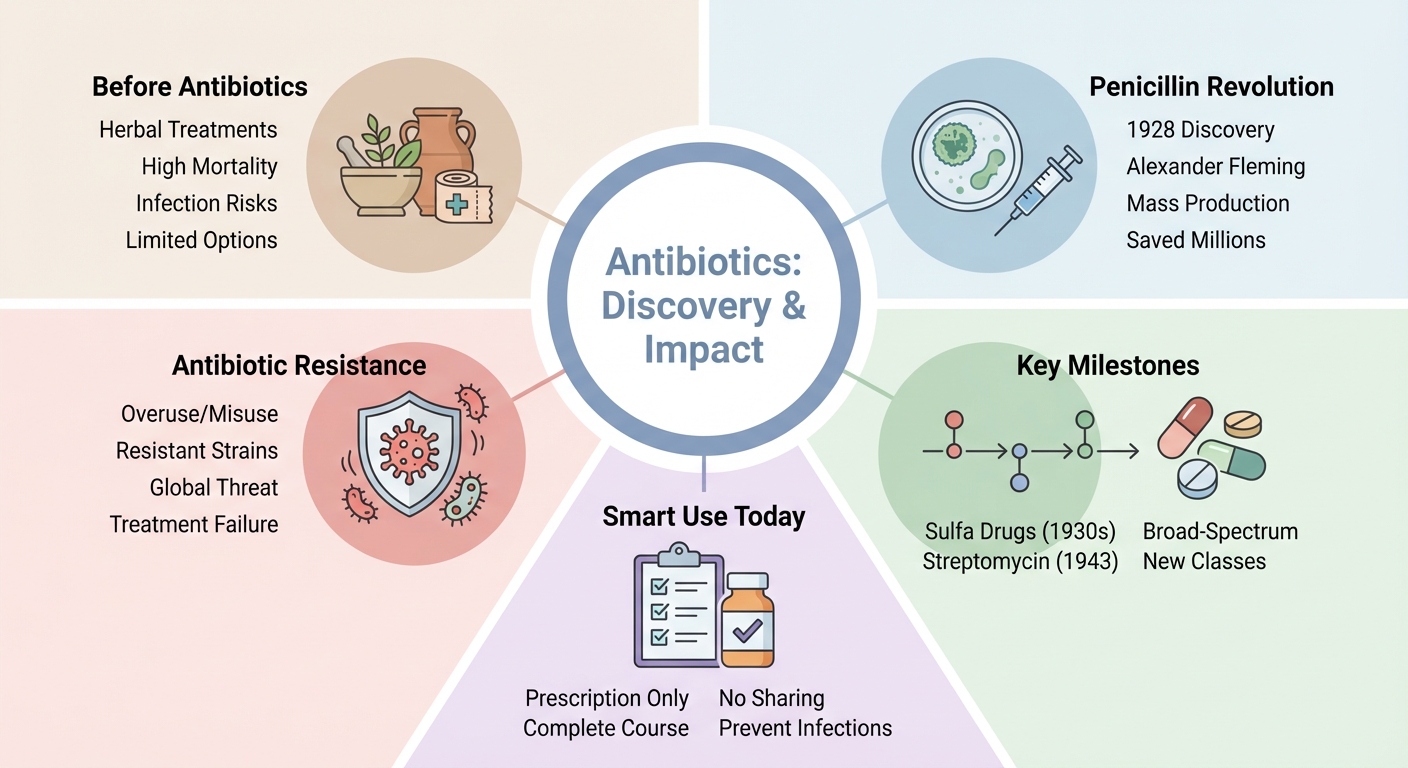
Alexander Fleming’s moment in 1928 – a Penicillium mold wiping out Staphylococcus bacteria on a petri dish – turned lab accident into international race.
The result was a sharp drop in deaths from pneumonia, sepsis, and infected wounds.
However, each successive antibiotic contributed to bacteria becoming more resilient. Resistance emerged almost immediately after cures appeared, ensuring the threat persisted from the moment the initial patient recovered and left the hospital.
Definition and origins of antibiotics
Antibiotics – chemicals that stop or kill bacteria – grew from the idea of antibiosis, where microbes compete and one knocks out another. Paul Vuillemin named it “antibiosis” in 1889, but Selman Waksman introduced “antibiotic” in 1942, marking it as a substance from microbes that stops bacteria.
Soil-dwelling bacteria, especially actinomycetes, were the first goldmine. Waksman and H. Boyd Woodruff isolated actinomycin in 1940, launching systematic antibiotic discovery.
Screening vast numbers of soil samples for antibacterial properties became routine, and novel drugs began appearing at an unprecedented pace. Antibiotics target only bacteria. Viruses don’t care about them.
What was the first antibiotic in history?
Penicillin came from Penicillium notatum mold and stands as the first real antibiotic. Fleming’s 1929 paper in the British Journal of Experimental Pathology set the field in motion.
Penicillin’s filtrate killed dangerous bacteria but didn’t harm animal cells. In February 1941, Albert Alexander at Oxford’s Radcliffe Infirmary received the first human dose – proof that it worked, though supply was tight.
Its ability to target bacterial cell walls made penicillin different from anything that came before.
Who is the father of all antibiotics?
Selman Waksman, born in Ukraine, is known as the father of antibiotics for defining and building the discipline. (5) He coined “antibiotic” and led the charge in screening, which produced streptomycin in 1943.
By 1954, Waksman established the Institute of Microbiology at Rutgers, supported by major pharmaceutical companies such as Merck. Earlier, Paul Ehrlich’s arsenic-based Salvarsan and the foundational work of Louis Pasteur and Robert Koch set the scene, but none of them established antibiotics as a separate field.
Waksman’s approach shifted drug discovery from chance to systematic, targeted search.
Pre-antibiotic era and early antimicrobial practices
Before the antibiotic era, medical care relied on chemical guessing, accidental breakthroughs, and last-ditch remedies. (4)
Gerhard Domagk tried Prontosil – an azo dye from Bayer – on mice with Streptococcus infections. The mice survived; those in the control group died. Hidden in Prontosil’s red dye was sulfanilamide, a compound the dye industry had used for years but hadn’t recognized as antibacterial.
Salvarsan, made by Alfred Bertheim and Paul Ehrlich, attacked Treponema pallidum with arsenic – sometimes curing syphilis, sometimes poisoning patients.
No drug before Prontosil or Salvarsan worked broadly or safely.
Domagk’s daughter Hildegard survived a nearly fatal infection in 1932 after taking Prontosil, cementing the drug’s place in medicine. Jumping from toxic chemicals to targeted agents was the shift that separated guesswork from design.
The first effective antibacterial agent was hidden for years in a common industrial dye.
| Agent | Discovery Method | Originator(s) | Year | Mechanism |
|---|---|---|---|---|
| Salvarsan | Systematic chemical modification | Paul Ehrlich, Alfred Bertheim | 1910 | Arsenic-based, targets Treponema pallidum |
| Prontosil | Empirical screening, animal testing | Gerhard Domagk | 1932 | Prodrug, releases sulfanilamide in vivo |
| Penicillin | Serendipity, mold contamination | Alexander Fleming | 1928 | Inhibits bacterial cell wall synthesis |
Prontosil’s track record in animals – and Domagk’s own family – forced pharmaceutical companies to revisit thousands of abandoned dyes, looking for hidden antibacterial action.
What did humans use before antibiotics?
Before antibiotics, infection control was a patchwork of plant extracts, minerals, and crude surgery – results varied, often fatally.
In the Dakhleh Oasis, human remains from ancient Sudanese Nubia show tetracycline in bone, likely from eating grain fermented with Streptomyces-contaminated soil. Ancient populations, it seems, took in natural antibiotics without knowing.
Egyptian and European doctors tried poultices, burning, and honey for wounds.
Joseph Lister started spraying carbolic acid in surgeries in 1865, kicking off chemical antisepsis. By the late 1800s, serum therapy – injecting animal antitoxins – cut deaths from diphtheria and tetanus, but only for certain infections.
Arsphenamine (Salvarsan), new in 1910, was the first targeted drug for syphilis, but its toxic effects limited its use. Every earlier method gave inconsistent, incomplete protection. Antibiotics soon changed the rules.
Golden age of antibiotic discovery from the 1940s to the 1960s
Drug factories were transformed by barrels of microbe cultures, and within less than twenty years, researchers discovered entirely new classes of antibiotics. (3)
Albert Schatz, working at Rutgers in 1943, pulled streptomycin from Streptomyces griseus, finally curing tuberculosis.
René Dubos had already discovered gramicidin and tyrocidin in 1939, and these were produced on a commercial scale.
Still, the flood of actinomycete-derived drugs defined the period.
Cephalosporins reached the clinic after Giuseppe Brotzu’s pivotal find in 1948, with Oxford’s Edward Abraham and Guy Newton developing the first versions.
Chloramphenicol – approved by the FDA in 1949 – offered broad-spectrum coverage, while erythromycin’s debut in 1952 expanded treatment options with the macrolide class.
The 1950s and 1960s were exceptional decades, with more classes of antibiotics introduced then than in any other period. After 1970, most new antibiotics came from modifying molecules found in this earlier era.
More new antibiotic classes appeared in two decades than in the entire century before or after.
Pharmaceutical chemistry in these years set the standard for today’s drug development process.
When were antibiotics invented and widely used?
Penicillin hit mass production during World War II, changing bacterial infection from a fatal certainty to a problem doctors could treat.
Howard Florey, Ernst Boris Chain, and Norman Heatley scaled up penicillin G, with British and American factories supplying the Allies by 1944.
The 1945 Nobel Prize in Physiology or Medicine went to them and Fleming for this achievement.
The 1943 discovery of streptomycin by Schatz and Waksman at Rutgers revolutionized tuberculosis treatment, earning Waksman the 1952 Nobel Prize.
By the close of the 1940s, penicillin and its analogs were standard in hospitals around the world, setting a new bar for infection treatment.
Antibiotic use in World War II was the starting gun for worldwide, standardized antibacterial treatment.
Evolution of antibiotic resistance over time
Now, across hospitals from Tokyo to São Paulo, multidrug-resistant bacteria kill more people each year than malaria or HIV. (2)
A global 2022 report counted 1.27 million deaths directly caused by bacterial antimicrobial resistance – outpacing even tuberculosis.
Resistance didn’t start with penicillin or sulfanilamide: integrons with resistance genes have circulated in environmental microbes for thousands of years.
Siberian permafrost holds resistance genes that predate human-made antibiotics by over 10,000 years.
Antibiotic resistance genes existed in ancient permafrost long before modern medicine invented antibiotics.
Staphylococcus found ways around Prontosil and sulfanilamide within a decade. By the 1960s, hospitals faced outbreaks of methicillin-resistant Staphylococcus aureus (MRSA) and vancomycin-resistant enterococci (VRE), forcing doctors to rotate through entire classes of drugs in one patient.
Later, carbapenem-resistant Enterobacterales (CRE) broke through the last line of carbapenems, making even routine infections impossible to treat.
Resistance genes move between species on plasmids and integrons, spreading faster than new antibiotics are released.
Alexander Fleming warned in his 1945 Nobel lecture that “the thoughtless person playing with penicillin is morally responsible for the death of the man who succumbs to infection with the penicillin-resistant organism.”
MRSA, VRE, and CRE now shape infection control in hospitals around the world, driving the need for fast diagnostics and isolation protocols.
The genetic arms race rages on in sewage plants, hospital wards, and the soil – steady, mostly invisible, and picking up speed.
| Bacterial Pathogen | Resistant Drug/Class | Clinical Impact |
|---|---|---|
| MRSA | Methicillin (and other β-lactams) | Severe hospital- and community-acquired infections; increased mortality and hospital stays |
| VRE | Vancomycin | Complicated bloodstream, urinary, and surgical site infections; limited therapeutic options |
| CRE | Carbapenems | High-mortality infections in ICUs; outbreaks with few or no effective antibiotics |
Bacterial resistance keeps outpacing antibiotic discovery, leaving medicine to play catch-up.
Challenges facing antibiotics today
Pharmaceutical companies – despite the rising threat – have scaled back antibiotic research. Bringing a new antibiotic to market can take ten years or more, and short treatment windows plus quick resistance mean profits vanish quickly.
Since 1970, only four new antibiotic classes have reached patients, while between 1940 and 1960, over a dozen made it through.
The World Health Organization found, as of 2021, 57 countries already had widespread resistance to last-resort drugs. Even so, many places, especially in sub-Saharan Africa and Southeast Asia, still lack basics.
Antibiotic stewardship is now required. Hospitals ration prescriptions, push de-escalation, and track resistance to keep antibiotics useful.
Without new drug incentives, stewardship is the last line – slowing resistance though it never stops it.
Doctors now face the strange reality where antibiotics are overprescribed in some places and missing in others where they’re needed most.
Comparing antibiotic classes across eras
Under a Japanese farm, actinomycetes produced streptomycin – setting off a chain of new antibiotic classes, from fermentation tanks to synthetic labs. (1)
Penicillins and cephalosporins, both beta-lactams, blocked bacteria from making cell walls, transforming treatment for pneumonia and sepsis by 1950.
Discovered in the late 1970s, carbapenems expanded this approach, overcoming bacteria resistant to other beta-lactams and enhancing ICU safety.
Amoxicillin came along as a more stable, easily absorbed penicillin and became the default for childhood infections worldwide.
The fully synthetic quinolones – think ciprofloxacin (Cipro), launched by Bayer in 1983 – skip fermentation.
Chemists built them to block bacterial DNA gyrase, stopping pathogens that penicillins couldn’t touch.
Linezolid, in the oxazolidinone group, reached the clinic in 2000 – one of the rare new classes in decades – by halting protein synthesis in tough staphylococci and enterococci.
Bactericidal antibiotics like amoxicillin and ciprofloxacin kill bacteria outright. Bacteriostatic agents such as linezolid stop bacteria from growing, leaving the immune system to finish the job.
Doctors pick drugs based on infection site, bacteria involved, and resistance: ciprofloxacin works for urinary tract infections from Escherichia coli, while linezolid is used for vancomycin-resistant enterococci in surgical wounds.
Antibiotic classes – natural, semi-synthetic, or fully synthetic – show how chemical, biological, and clinical priorities have shifted over time.
Sources
- Rutgers University states streptomycin came from Streptomyces griseus discovered at the New Jersey Agricultural Experiment Station in 1943. Separately, standard pharmacology references classify penicillins and cephalosporins as beta-lactams that inhibit bacterial cell-wall synthesis – rutgers.edu
- WHO briefing citing the 2022 Lancet systematic analysis of bacterial AMR in 2019; WHO states 1.27 million deaths per year were directly caused by bacterial AMR and 4.95 million were associated with bacterial AMR, which is more than HIV, TB, and malaria combined – apps.who.int
- Rutgers University news article on the discovery of streptomycin; it states that in 1943 at the New Jersey Agricultural Experiment Station, Selman Waksman with graduate students Albert Schatz and Elizabeth Bugie found that Streptomyces griseus produces streptomycin – rutgers.edu
- Nobel Prize presentation speech for Gerhard Domagk; it states Prontosil was tested in mice infected with haemolytic streptococci, that the work was directed by Domagk, and that the earliest published experiments began in December 1932. The claim’s Bayer reference is consistent with Bayer’s IG Farben context, though the Nobel page names the Igefa laboratory rather than Bayer directly – nobelprize.org
- Rutgers University profile on Selman Waksman; Rutgers states Waksman coined the term ‘antibiotics’ and credits him and Albert Schatz with the discovery of streptomycin – ucmweb.rutgers.edu
F.A.Q
Who discovered the first antibiotic?
Alexander Fleming discovered penicillin, the first antibiotic, in 1928 after observing mold in a petri dish that killed bacteria.
What did people use to treat infections before antibiotics?
Prior to antibiotics, treatments for infections included plant extracts, minerals, basic surgery, and chemical antiseptics such as carbolic acid.
What is the difference between antibiotics and other antimicrobial treatments?
Antibiotics specifically target and kill bacteria, while other antimicrobial treatments may use chemicals or methods that are less precise or more toxic.
Why did antibiotic resistance appear so quickly after antibiotics were introduced?
Resistance in bacteria emerged soon after antibiotics were introduced, as bacteria quickly adapted to survive drugs intended to kill them.
Who is considered the father of antibiotics?
Selman Waksman is known as the father of antibiotics because he coined the term and led efforts to discover new antibiotic drugs.
What was the first antibiotic used in humans?
Penicillin was the first antibiotic used in humans, with the initial patient treated at Oxford's Radcliffe Infirmary in 1941.
How have antibiotics changed over time?
Originally derived from natural molds like penicillin, antibiotics now include fully synthetic types, with new drugs developed to fight resistant bacteria and treat more infections.